/halogens-56a12cdc3df78cf772682689.png)
This reactivity means you’ll never find pure elemental fluorine in nature. No chemical on the planet can separate fluorine from any of its compounds. It also refuses to let go of any of its ill-gotten gains. It’s the most reactive element on the periodic table, bonding with anything that gets close enough to swap electrons. Fluorineįluorine is a bit of an elemental problem child. Where can you find the halogen elements in your daily life? Since the nature of halogen properties makes then hard to find alone, here are common diatomic molecules of each element found in our lives. Tennessine is a bit difficult to classify because it was created in a lab, so there have only been a few atoms created at any given time, and the element has a half-life of only around 80 milliseconds. Iodine and astatine are found as solids, bromine is found as a liquid, and fluorine and chlorine in their natural state are gasses. Unlike some of the other elemental groups, halogens span all three states of matter. They’re all found as diatomic molecules - two atoms of the element - or bonded with another element.Įach of these elements will also form acids when combined with hydrogen. In their pure form, you’ll never find a single atom of a halogen element. Halogen properties are identified by their outer shell - each halogen element has seven valence electrons in its outer shell. Chemical and Physical TraitsĮach of these elements has similar chemical and physical properties. 
On the other hand, scientists artificially created Tennessine. Scientists consider astatine one of the rarest naturally occurring elements. The six are: Flourineįluorine and chlorine are fairly abundant in the Earth’s crust. 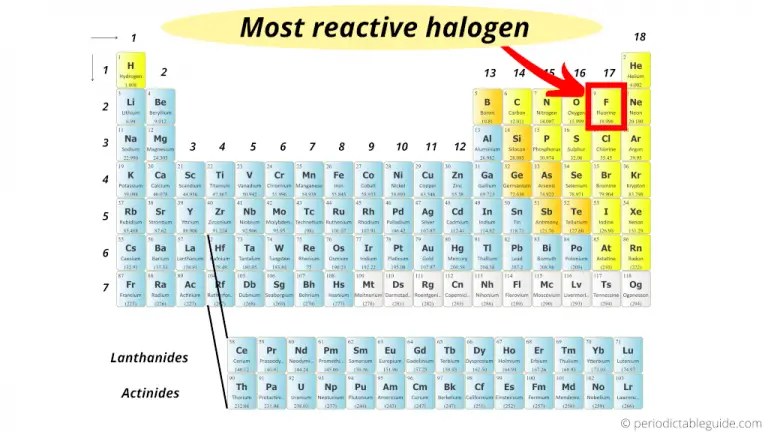
These elements are very reactive, so when they’re found in nature, it’s usually in the form of compounds or as ions. There are six elements in the halogen group, but they’re very rarely found in their pure form in nature. What are halogens, and where might you encounter them in your daily life? Halogen Properties and Order of Abundance Today, we’re going to take a closer look at halogen properties. The various metals make up everything from the silverware you eat with to pieces of the car you drive. Oxygen and nitrogen make up the air you breathe. Oxygen and hydrogen make up the water you drink. We use the different elements of the periodic table every single day without even realizing it. When you buy through links on our site, we may earn an affiliate commision. Some halides of the world are a part of molecules with covalent bonds.Revolutionized is reader-supported. Don't think that the halogens always make ionic compounds and salts. One of the best examples of a halide is sodium chloride (NaCl). When a halogen combines with another element, the resulting compound is called a halide. The elements we are talking about in this section are called halogens. Their chemical properties change just a little bit when compared to the element right above them on the table. As the atomic number increases, the atoms get bigger. As you learn more about the table, you will find this pattern true for other families. Reactivity decreases as you move down the column. Fluorine is the most reactive and combines with most elements from around the periodic table. Not all halogens react with the same intensity or enthusiasm. 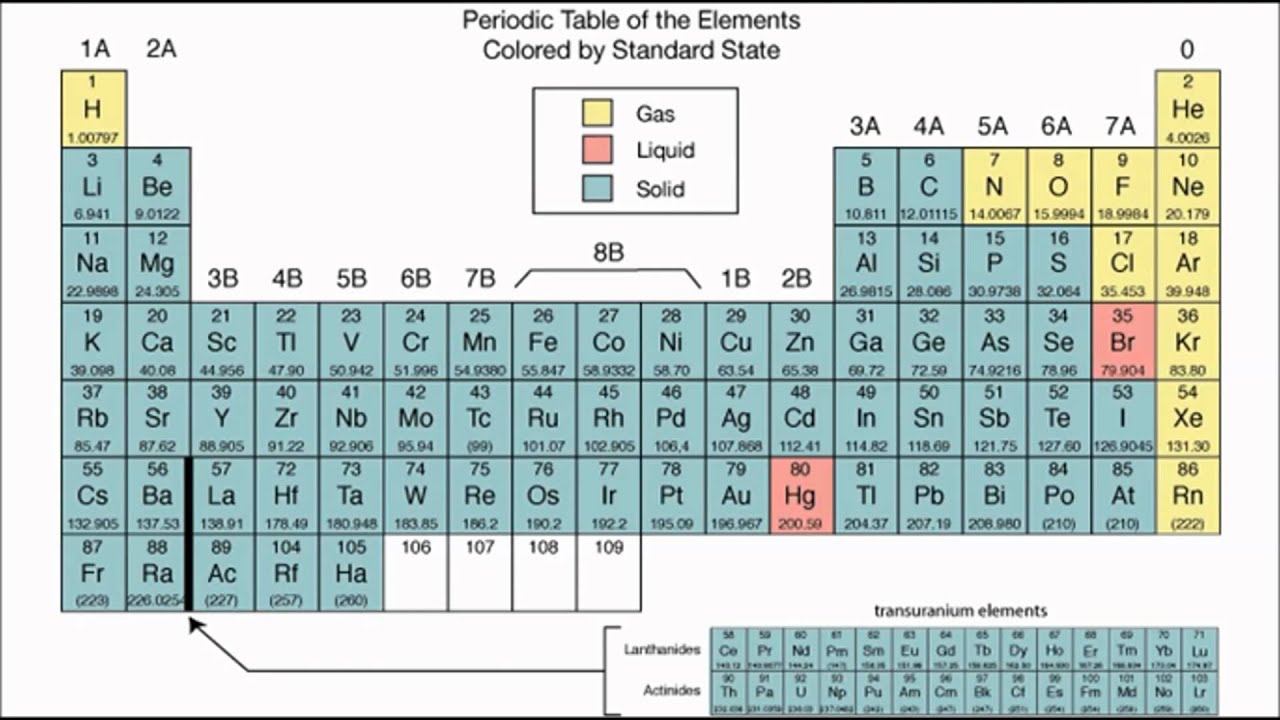
We've just told you how reactive the halogens are. The elements in the column on the left each have one electron that they like to donate. You will often find them bonding with metals and elements from Group One of the periodic table. Because they are so close to being happy, they have the trait of combining with many different elements. They are all just one electron shy of having full shells. That seven-electron trait applies to all of the halogens. When you look at our descriptions of the elements fluorine and chlorine, you will see that they both have seven electrons in their outer shell. Who is in this family? The elements included are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). This column is the home of the halogen family of elements. In the second column from the right side of the periodic table, you will find Group Seventeen (Group XVII).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
